Evolution in computational interface physics
- Kolloquium
Evolution in computational interface Physics
Liquid interfaces are not just a fascinating subject in the realm of nature; they also hold profound implications across a broad spectrum of scientific and technological fields. Their significance stems from their nanometer-scale dimensions and their inherently dynamic, disordered nature, which is characterized by weak intermolecular interactions at the order of kBT. Consequently, the study of these interfaces through experimental methods presents a formidable challenge.
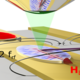
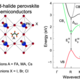
In this presentation, I aim to captivate the audience with the enigmatic behavior and physics underlying liquid interfaces, with a special emphasis on liquid crystalline interfaces such as lipid bilayers, which emerge through a process known as surfactant self-assembly [1]. The latter part of the talk will delve into how biomolecules can detect alterations in liquid interface properties, including curvature, molecular composition, and liquid order. I will illustrate how the integration of computational interface physics using coarse-grained molecular simulations, evolutionary algorithms, and machine learning techniques is now rapidly advancing our ability in deciphering amino acids sequences that can sense distinct interface properties such as curvature [2], molecular composition, and order.
I will illustrate how these advancements have now led to the discovery of potent antiviral peptide sequences that specifically target the liquid interface of small enveloped viruses. This breakthrough marks a significant leap forward in antiviral research. Additionally, our latest research has uncovered the first liquid-ordered phase binding sequence. These sequences are hypothesized to be pivotal in the recognition of liquid ordered domains potentially present in cellular membranes, a concept that has long been a subject of speculation. Finally, I will
delve into how the identified principles underlying curvature sensing are alternatively exploited to enhance the transport of medications across the blood-brain barrier [3].
[1] Soleimani, A., & Risselada, H. J. (2023). Pure Graphene Acts as an “Entropic Surfactant” at the Octanol–Water Interface. ACS nano, 17(14), 13554-13562.
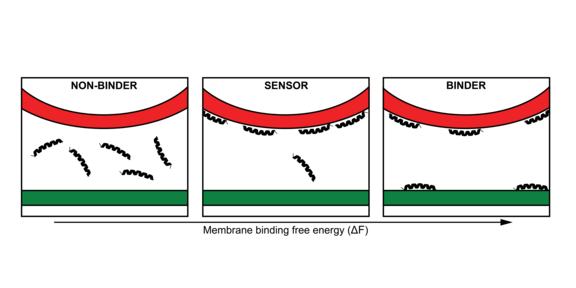
[2] van Hilten, N., Methorst, J., Verwei, N., & Risselada, H. J. (2023). Physics-based generative model of curvature sensing peptides; distinguishing sensors from binders. Science Advances, 9(11), eade8839.
[3] Papadopoulou, Panagiota, et al. "Phase‐Separated Lipid‐Based Nanoparticles: Selective Behavior at the Nano‐Bio Interface." Advanced Materials 36.6 (2024): 2310872.